Bacteriophages against mycobacteria, phage therapy for tuberculosis
Mycobacterial infections pose a serious challenge to modern medicine, particularly in the fight against Mycobacterium tuberculosis, the causative agent of tuberculosis —multidrug-resistant (MDR) and extensively drug-resistant (XDR) strains. The world is searching for new, alternative antibiotic treatments for tuberculosis, and bacteriophages are considered a very promising approach.
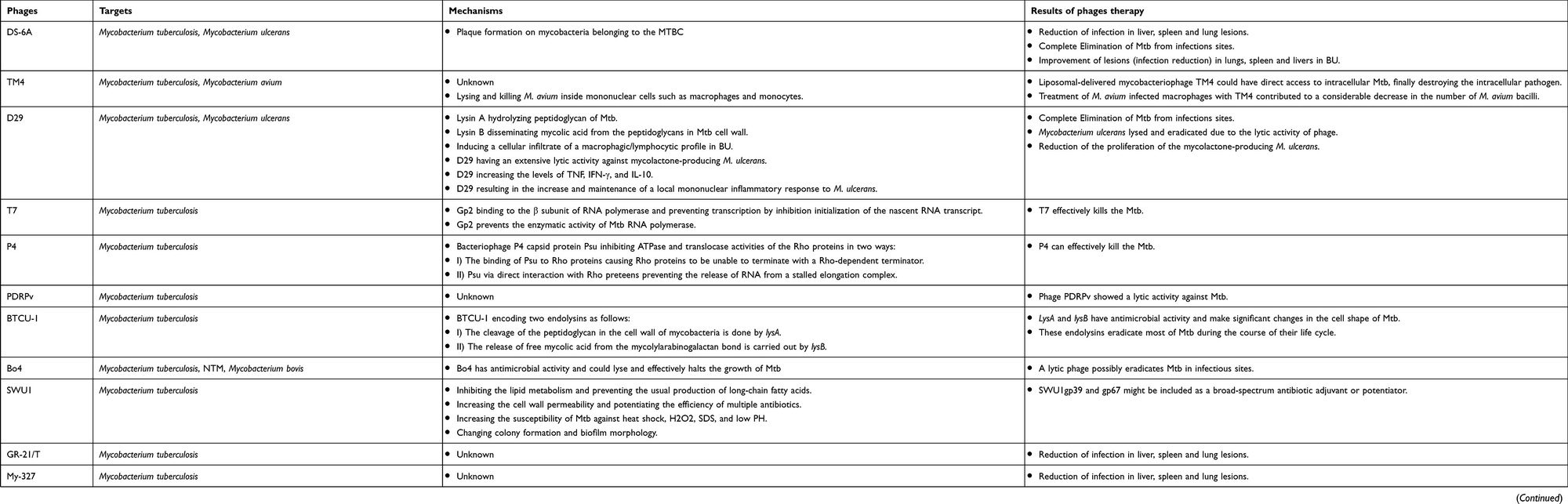
The ability of bacteriophages to destroy mycobacteria in vitro has been well studied, but data on phage therapy in humans for mycobacterial infections in general and tuberculosis in particular is scarce. Current data on this topic are summarized in a review by Iranian researchers led by Taher Azimi* and presented in Table*.
A separate issue highlighted by the authors* is that mycobacteria are intracellular parasites, meaning they live inside their host cells. Therefore, they are resistant to most antibiotics and are inaccessible to bacteriophages. Therefore, implementing phage therapy for mycobacterial infections requires solving the problem of delivering bacteriophages into the host cell. Several options are currently being considered.
The first and highly promising method for delivering phages to human cells is the use of mycobacteria themselves, specifically the non-virulent, fast-growing species Mycobacterium smegmatis . It does not cause disease even in people with suppressed immune systems. In phage therapy, M. smegmatis can perform several functions: 1) serve as a carrier that delivers bacteriophages within itself into host cells, where the phages then release and infect virulent mycobacteria; 2) act as a target bacterium, ensuring rapid growth of bacteriophages; 3) increase the rate of infection of pathogens by bacteriophages; 4) serve as a medium for bacteriophage activity within macrophages and monocytes.
Another possible method for delivering phages to target pathogenic mycobacteria involves the use of liposomes. Liposomes are vesicles composed of a lipid bilayer similar in composition and structure to the cell membrane. They fuse with the membrane of eukaryotic cells and, via endocytosis, are delivered along with their contents into the cytoplasm of the cells. In an experiment, bacteriophage TM4, specific for M. tuberculosis , was packaged in liposomes and added to a culture of cells infected with the tuberculosis pathogen. After fusion with the cells, the bacteriophages destroyed the pathogenic bacteria, indicating that the liposomes successfully delivered the therapeutic payload to its destination.
* Azimi T, Mosadegh M, Nasiri MJ et al. Phage therapy as a renewed therapeutic approach to mycobacterial infections: a comprehensive review // Infection and Drug Resistance, 2019, 12: 2943-2959. DOI https://doi.org/10.2147/IDR.S218638